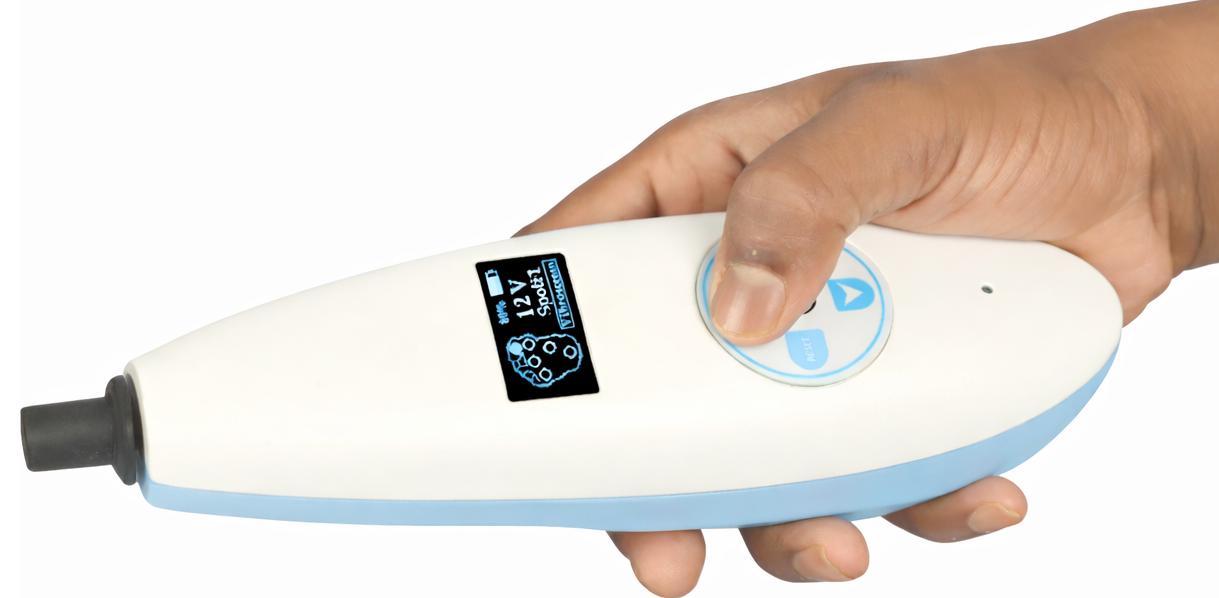
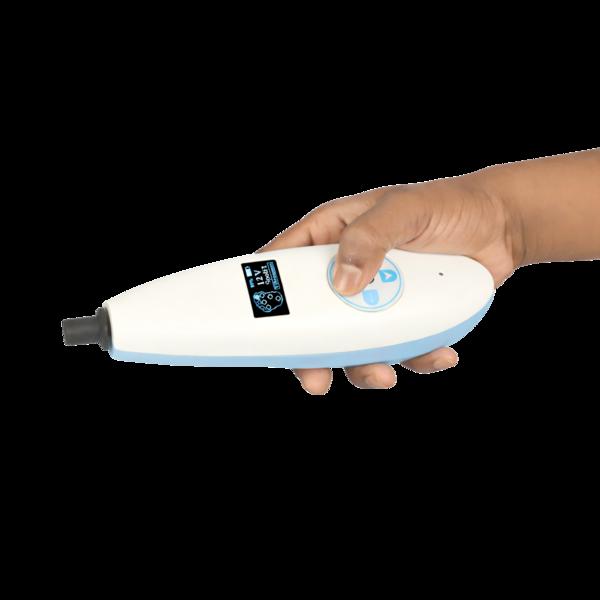
VIBROSCREEN is a next-generation, handheld Biothesiometer engineered for objective assessment of vibration perception threshold (VPT) in patients at risk of Diabetic Peripheral Neuropathy. The device is optimized for point-of-care diagnostics, enabling rapid, reproducible, and operator-independent screening across clinical, community, and domiciliary settings. Its compact, self-contained architecture, combined with digital integration capabilities, supports large-scale neuropathy screening programs and longitudinal patient monitoring. Technical & Functional Specifications: 1. Handheld, Portable Architecture • Compact, lightweight, and ergonomically engineered for single-handed operation • Suitable for bedside evaluation, outpatient clinics, screening camps, and home healthcare delivery • Battery-powered system ensuring complete operational mobility without dependence on mains supply • Probe geometry optimized for accurate application on plantar surfaces 2. Vibration Stimulus Generation • Employs a precision-controlled electromechanical probe for vibration delivery • Operates at a fixed frequency of 128 Hz, aligned with standard clinical protocols for VPT assessment • Factory-calibrated output ensures high reproducibility and inter-device consistency • Designed to stimulate large myelinated Aβ fibers, facilitating early detection of sensory neuropathy 3. Adjustable Stimulus Amplitude (Voltage-Controlled) • Vibration amplitude is directly proportional to applied voltage, enabling graded stimulus delivery • Continuous and incremental voltage modulation allows accurate determination of perception thresholds • Measurement range typically spans 0–30 V, compliant with established clinical screening thresholds • Supports quantitative sensory testing methodology for neuropathy grading 4. Digital Display & User Interface • Integrated high-contrast OLED display for real-time visualization of VPT values • Displays stimulus intensity (Volts) with high resolution for precise documentation • User-friendly interface minimizes operator training requirements and reduces variability 5. Threshold Measurement & Operating Modes • Manual Mode: o Clinician-controlled application of vibration stimulus at standard anatomical sites o VPT values recorded manually in structured clinical documentation formats • Computer/Mobile-Assisted Mode: o Device interfaces with Windows-based systems or Android platforms o Automated test workflows with guided protocols and standardized site sequencing o Real-time data acquisition, storage, and algorithm-based interpretation o Fully functional in offline environments, ensuring usability in low-resource settings 6. Power Management System • Integrated rechargeable lithium-ion battery with USB Type-C charging interface • Optimized for extended field use with low power consumption architecture • Capable of screening approximately 150–200 patients per full charge cycle • Includes battery status monitoring for uninterrupted clinical workflow 7. Smart Connectivity & Data Integration • Embedded Bluetooth Low Energy (BLE) module for seamless wireless data transfer • Enables integration with mobile and desktop applications for: o Patient data management o Longitudinal tracking and trend analysis o Digital report generation • Supports combined neuropathy screening with: o 10 g Semmes-Weinstein monofilament testing (protective sensation assessment) • Designed for interoperability within a broader diagnostic ecosystem, including: o Ankle-Brachial Index (ABI) systems o Plantar pressure distribution and foot risk assessment platforms Clinical Relevance VIBROSCREEN facilitates early identification of loss of protective sensation (LOPS), a critical risk factor for diabetic foot ulceration and amputation. Its deployment in primary care and screening programs enhances preventive foot care strategies and supports evidence-based clinical decision-making.

About Diabetik Foot Care India Private Limited
We care for Footcare
28
HAPPY CLIENTS
2
SKILLED STAFF
Have any custom requirements?
Testimonials
View All
All India Institute of Medical Sciences, Nagpur
Working Satisfactorily, and the performance of the equipments as well as the services provided by the vendor have been f...
Read MoreDr Anoop Misra

Testimonial
Diabetes Foundation New Delhi
Dr Vijay Viswanathan

Testimonial
M V Hospital for Diabetes
×
![]()